The “-2” standard is increasingly important in the electrical medical device world.
Dan Hoolihan, Houlihan EMC Consulting, Lindstrom, MN, USA
INTRODUCTION
The most well-known and used EMC Standard for Electrical Medical Devices is IEC 60601-1-2 (“-2”) which is entitled “Medical Electrical Equipment – Part 1-2: General Requirements for Basic Safety and Essential Performance – Collateral Standard: Electromagnetic Compatibility – Requirements and Tests.” The standard was most recently released as the Third Edition in March of 2007. The Standard is currently under review by Maintenance Team 23 of Subcommittee 62A (Common Aspects of Electrical Equipment Used in Medical Practice), which is part of the International Electrotechnical Committee – Technical Committee 62 (Electrical Equipment in Medical Practice).
This article discusses some of the changes from Edition 2 to Edition 3, reviews key-requirements of the “-2” standard, and discusses possible future directions for the standard. It is an “overview” type of article since the actual standard is too long and detailed to review in depth.
CHANGES FROM EDITION 2.0 TO EDITION 3.0
In the Foreword of the third edition, it is stated “This edition of the IEC 60601-1-2 was revised to structurally align it with the 2005 edition of IEC 60601-1 and to implement the decision of IEC subcommittee 62A that the clause numbering structure of collateral standards written to IEC 60601-1:2005 would adhere to the form specified in ISO/IEC Directives, Part 2:2004. The principal technical changes are in Clause 4, which now recognizes that there is a general requirement for a risk management process in IEC 60601-1:2005.”
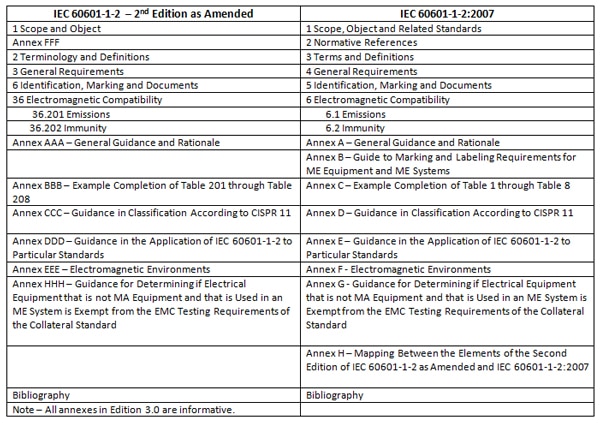
Additional changes include a new Clause 1.3 entitled Related Standards, a new Annex B (Guide to Marking and Labeling Requirements for Medical Electrical [ME] Equipment and Medical Electrical [ME] Systems), and Annex H (Mapping between the Elements of the Second Edition of IEC 60601-1-2 as amended and IEC 60601-1-2:2007).
Clause 1.3 of the “-2” standard has two sub-clauses; the first is titled “IEC 60601-1” and basically says that “this collateral standard complements IEC 60601-1.” The second sub-clause is “Particular Standards.” It states that “A requirement in a particular standard takes priority over the corresponding requirement in this collateral standard.”
Annex B is Informative and has two sub-clauses; B.1 (Marking on the Outside of ME Equipment, ME Systems, or Their Parts) and B.2 (Accompanying Documents, Instructions for Use). Both sub-clauses refer to specific sub-clauses in the body of the standard for the reader’s guidance.Annex H does an excellent job of mapping the requirements between Edition 2 and Edition 3 of the “-2” standard. An abbreviated version of Annex H is shown in Table 1.
TESTING REQUIREMENTS – EMISSION
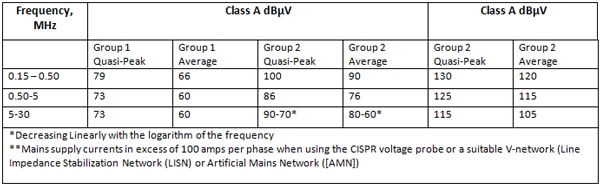
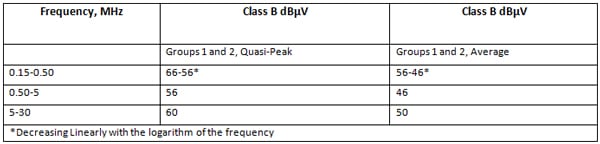
ME Equipment and ME Systems are classified as either Class A or Class B and Group 1 or Group 2 as per CISPR 11 (Industrial, Scientific and Medical (ISM) Radio-Frequency Equipment – Electromagnetic Disturbance Characteristics – Limits and Methods of Measurement).
Class A equipment is equipment suitable for use in all establishments other than domestic and those directly connected to the public low-voltage power supply network that supplies buildings used for domestic purposes
Class B equipment is equipment suitable for use in all establishments including domestic establishments and those directly connected to the public low-voltage power supply network that supplies buildings used for domestic purposes
Group 1 equipment contains all ISM equipment in which there is intentionally generated or used conductively-coupled Radio Frequency (RF) energy that is necessary for the internal functioning of the equipment itself
Group 2 equipment contains all ISM equipment in which RF energy is intentionally generated or used in the form of electromagnetic radiation for the treatment of material, and spark erosion equipment
Most medical equipment is Group 1 equipment.
- Some examples of Group 2 equipment are:
- Medical Imaging Equipment (systems for magnetic resonance imaging)
- Therapy Equipment (diathermy equipment including short-wave, ultra-short-wave, and microwave)
- Therapy Equipment (hyperthermy equipment)
- High-Frequency Surgical Equipment and Systems (because they apply RF energy to the patient)
CONDUCTED EMISSION REQUIREMENTS
It can be seen that the Class B limits for Group 1 equipment are anywhere from 10 to 17 dB lower than the Class A limits depending on the frequency range.
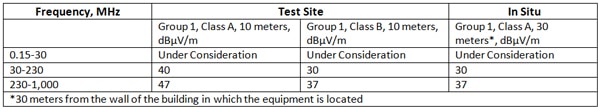
There are additional tables of emission limits in CISPR 11 which cover (1) Mains Terminal Disturbance Voltage for Induction Cooking Appliances; (2) Limits of the Magnetic Field Induced Current in a two-meter loop antenna around the device under test; (3) limits of the magnetic field strength; (4) Electromagnetic Radiation Disturbance Limits for Group 2, Class B Equipment Measured on a Test Site; (5) Electromagnetic Radiation Disturbance Limits for Group 2, Class A Equipment; (6) Electromagnetic Radiation Disturbance Limits for Class A Electro-Discharge Machining (EDM) and Arc Welding Equipment Measured on a Test Site; (7) Electromagnetic Radiation Disturbance Peak Limits for Group 2, Class A and Class B ISM Equipment Producing CW-type Disturbances and Operating at Frequencies above 400 MHz, (7) Electromagnetic Disturbance Peak Limits for Group 2, Class B ISM Equipment Producing Fluctuating Disturbances other than Continuous Wave (CW) and Operating at Frequencies above 400 MHz; (8) Electromagnetic Radiation Disturbance Weighted Limits for Group 2, Class B ISM Equipment Operating at Frequencies above 400 MHz; and (9) Limits for Electromagnetic Radiation Disturbances to Protect Specific Safety Services in Particular Areas.
ME equipment and ME systems with a rated input current of less than 16 amps per phase must comply with IEC 61000-3-2 ( EMC – Part 3-2: Limits – Limits for Harmonic Current Emissions) for harmonic distortion.
ME equipment and ME systems with a rated input current of less than 16 amps per phase must also comply with IEC 61000-3-3 (EMC – Part 3-3: Limits – Limitation of Voltage Changes, Voltage Fluctuations and Flicker in Public Low-voltage Supply Systems, for equipment not subject to conditional connection) for voltage fluctuations and flicker.
Both IEC 61000-3-2 and IEC 61000-3-3 specify limits, test methods, and test equipment.
IMMUNITY
The Immunity requirements of “-2” are based on the International Immunity Standards contained in the series of IEC 61000-4-x standards where x is 2, 3, 4, 5, 6, and 11.
The Electrostatic Discharge (ESD) criteria are contained in IEC 61000-4-2 (EMC – Part 4-2: Testing and Measurement Techniques – Electrostatic Discharge Immunity Test). The requirements are that the medical equipment shall comply with test levels of ±2 kV, ±4 kV, and ±8 kV for air discharge and ±2 kV, ±4 kV, and ±6 kV for contact discharge.
The Radiated RF Electromagnetic Field criteria are taken from IEC 61000-4-3 (EMC – Part 4-3: Testing and Measurement Techniques – Radiated, Radio-Frequency, Electromagnetic Field Immunity Test). The test requirements are 3 volts per meter over the frequency range of 80 MHz to 2.5 GHz for Non-Life-Supporting equipment and 10 volts per meter over the same frequency range for Life-Supporting equipment.
For Electrical Fast Transients and Bursts, medical equipment shall have an immunity test level of plus or minus 2 kilovolts for alternating current and direct current power lines and plus or minus 1 kilovolt for signal and interconnecting cables. The test method shall be as per IEC 61000-4-4 (EMC – Part 4-4: Testing and Measurement Techniques – Electrical Fast Transient/Burst Immunity Test) which simulates “electrical noise” from switching relays.
Surge testing is also covered in IEC 60601-1-2, and the testing is done as per IEC 61000-4-5 (EMC – Part 4-5: Testing and Measurement Techniques – Surge Immunity Test). The requirements to be met are test levels of ±0.5 kV, ±1 kV, and ±2 kV for alternating current power lines to ground and ±0.5 kV and ±1 kV for alternating current phase-to-phase voltage testing. This test simulates secondary effects of lightning discharges.
The next immunity test is for conducted disturbances induced by RF fields as per IEC 61000-4-6 (EMC – Part 4-6: Testing and Measurement Techniques – Immunity to Conducted Disturbances, Induced by Radio-Frequency Fields). The test level requirement shall be 3 volts (rms) over the frequency range of 150 kHz to 80 MHz for Non-Life-Supporting Equipment. The RF signal shall be modulated at either 2 Hz or 1000 Hz depending on the intended use of the equipment under test. Life-Supporting Equipment shall meet 3 volts (rms) over the frequency range of 150 kHz to 80 MHz except at the ISM frequency bands in that range where the EUT must meet 10 volts (rms). This test simulates cables acting as receiving antennas and converting radiated fields into conducted RF signals on the cables.
The last immunity test is a test for Voltage Dips, Short Interruptions, and Voltage Variations on Power Supply Input Lines. The appropriate standard is IEC 61000-4-11 (EMC – Part 4-11: Testing and Measurement Techniques – Voltage Dips, Short Interruptions, and Voltage Variation Immunity Tests). The criteria to be met are summarized in Table 10 of “-2.” Equipment and systems that are not life-supporting and that draw more than 16 amps per phase– are exempt from this testing. Non-Life-Supporting Equipment and Systems with a rated input power of 1 kVA or less and all Life-Supporting Equipment and Systems shall comply with the criteria in Table 10. Some exceptions to the requirements are allowed for Non-Life-Supporting equipment that draws more than 1 kVA of power but less than 16 amps per phase. This is a test that simulates everyday power fluctuations in the input alternating current power lines that feed the power supplies of the medical device equipment.
NON-TESTING REQUIREMENTS OF IEC 60601-1-2
Non-testing requirements of the standard are covered in Clause 5 of Edition 3.0 and include identification, marking, and accompanying documents.
Identification and Marking
ME equipment and ME systems that include RF transmitters or that intentionally apply RF electromagnetic energy for diagnosis or treatment shall be labeled with the following symbol for non-ionizing radiation:
For ME equipment and ME systems for which the connector testing exemption specified in 6.2.2.2 c) is used, symbol IEC 60147-5134 (2003-04) for ESD sensitivity shall be applied adjacent to each connector for which the testing exemption is used.
ME equipment and ME systems specified for use only in a shielded location shall be labeled with a warning that they should be used only in the specified type of shielded location.
Accompanying Documents
In the Instructions for Use, all ME equipment and ME systems shall include the following:
- A statement that medical electrical equipment needs special precautions regarding EMC and needs to be installed and put into service according to the EMC information provided in the accompanying documents; and
- A statement that portable and mobile RF communications equipment can affect medical electrical equipment
In the Technical Description, the accompanying documents for all ME equipment and ME systems shall include the following:
- A list of all cables and maximum lengths of cables (if applicable), transducers, and other accessories with which the manufacturer of the ME equipment and ME system claims compliance with the requirements of 6.1 and 6.2. Accessories that do not affect compliance with the requirements of these subclauses need not be listed. Accessories, transducers, and cables may be specified either generically (e.g., shielded serial cable, load impedance) or specifically (e.g., by manufacturer and model or part number).
- A warning that the use of accessories, transducers, and cables other than those specified, with the exception of transducers and cables sold by the manufacturer of the equipment or system as replacement parts for internal components, may result in increased emissions or decreased immunity of the ME equipment or ME system.
- A warning that the ME equipment or ME system should not be used adjacent to or stacked with other equipment and that if adjacent or stacked use is necessary, the ME equipment or ME system should be observed to verify normal operation in the configuration in which it will be used.
- A justification for each compliance level that is lower than the IEC 60601 test level for that immunity test. These justifications shall be based only on physical, technological, or physiological limitations that prevent compliance with the IEC 60601-1 test level.
- Table 2 completed as per the flowchart of Figure 3.
- The performance of the ME equipment or ME system that was determined to be Essential Performance.
Testing for Safety vs. Testing for Performance
The activities of the Maintenance Team working on the fourth Edition of IEC 60601-1-2 are centered around the issue of separating the standard into two standards; one for Safety (EMC and Safety are considered separate concepts) and one for EMC for Performance. This is a very difficult concept to implement in the standard.
The major difference would be that for a Safety standard a medical device could fail an immunity test as long as it “failed safely.” That is, if the equipment shut down yet did not harm the patient in any way (did not create an unacceptable risk), that could be an acceptable outcome.
However, in the EMC Performance standard, if the medical device were to fail an immunity test at a “standard” level of electromagnetic influence, that medical device would have to be redesigned so that it could pass the minimum “standard” level of electromagnetic energy specified in the test.
CONCLUSION
The “-2” standard is increasingly important in the electrical medical device world. Equipment that complies with the requirements of the standard has the potential of being used seamlessly around the world. The concept of EMC for Safety and EMC for Performance is being debated relative to the next edition of the standard. Stay tuned as the Maintenance Team for the international standard discusses the multitude of technical and administrative issues relative to the Safety/Performance split.







